Your Trusted Resource for Evidence-Based Information on Lyme and other Tick-Borne Illnesses
Because quality science is the best medicine

Why ALDF?

Scientifically Accurate Information
We are committed to providing accurate and reliable information backed by scientific research. Our mission is to dispel myths and misconceptions surrounding Lyme disease and other tick-related illnesses.

Credible, Transparent Resources You Can Rely On
We meticulously screen the information we share to ensure that it is trustworthy and provided by reliable sources. With a focus on transparency, we provide you with provide you with science-based facts about Lyme disease and other tick-related illnesses.

Complete coverage: Prevention, Diagnosis, and Treatment of Lyme disease
ALDF provides comprehensive and reliable information on Lyme disease, covering everything from prevention to diagnosis and treatment. With a commitment to research, education, and prevention, we strive to be a useful and holistic resource to our readers.

Decades of Pioneering and Leading Lyme Disease Education
Our organization consists of a diverse group of expert health professionals and researchers, each bringing years of invaluable knowledge and experience. We have been dedicated to advancing Lyme disease education for physicians and the public since 1990.

Tribute to Phil Baker
The Best Available Scientific Evidence Published
in Medical Literature
Lyme Disease Resources
With our reliable resources, we empower physicians to make informed decisions and contribute to the prevention and management of Lyme disease. Join us in our mission to research, educate, and prevent Lyme disease. The future of healthcare starts with you.
Lyme Disease Prevention, Diagnosis, and Treatment
Lyme disease is a tick-borne illness caused by the bacteria Borrelia burgdorferi. It can lead to a wide range of symptoms, including fatigue, joint pain, and neurological problems. Early diagnosis is crucial for effective treatment. Diagnosis is typically based on symptoms, medical history, and a blood test to detect antibodies against B. burgdorferi. Early treatment can help prevent complications and long-term health issues.
Chronic Lyme Disease
We caution patients against seeking providers who use treatment regimens different from the ISDA guidelines and those who use non-standard or experimental Lyme disease tests.


Prevention

Diagnosis

Treatment

Chronic Lyme Disease

Tick Species

Tick-Borne Illnesses

Updates in Lyme Research

SUPPORT OUR CAUSE
Risk of Contracting Lyme Disease by State
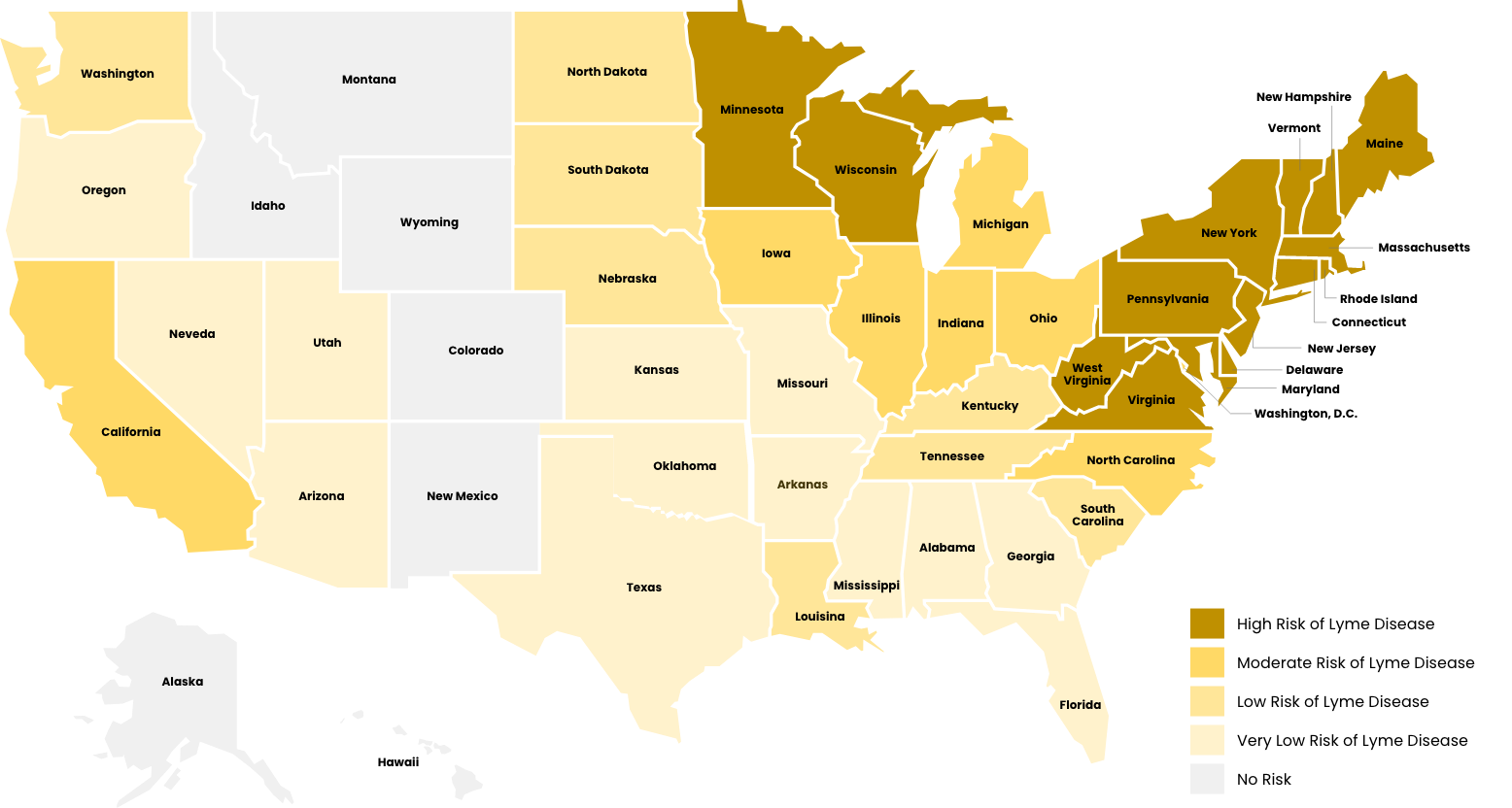
- High Risk of Lyme Disease
- Moderate Risk of Lyme Disease
- Low Risk of Lyme Disease
- Very Low Risk of Lyme Disease
- No Risk of Lyme Disease
High risk areas, relevant Ixodes tick species are widespread and have been found to be consistently infected with Borrelia burgdorferi.
- Pennsylvania
- New Jersey
- New York
- Wisconsin
- Maine
- New Hampshire
- Minnesota
- Maryland
- Connecticut
- Virginia
- Vermont
- Rhode Island
- West Virginia
- Massachusetts
- Delaware
*Risk is calculated by prevalence/existence of either Ixodes scapularis or Ixodes pacificus ticks, the bacterium (Borrelia burgdorferi spp.), epidemiological data demonstrating ticks are infected, and population size/distribution in areas where people are found.
Moderate risk areas, relevant Ixodes tick species can be found infected with Borrelia burgdorferi, but distribution across the state is less widespread or clustered in specific areas.
- California
- Ohio
- Michigan
- Illinois
- North Carolina
- District of Columbia
- Iowa
- Indiana
*Risk is calculated by prevalence/existence of either Ixodes scapularis or Ixodes pacificus ticks, the bacterium (Borrelia burgdorferi spp), epidemiological data demonstrating ticks are infected, and population size/distribution in areas where people are found.
Low risk areas, relevant Ixodes species have been found, but only sparse areas where they have been identified to be carrying Borrelia burgdorferi.
- Tennessee
- Kentucky
- South Carolina
- North Dakota
- Washington
- South Dakota
- Nebraska
- Louisiana
*Risk is calculated by prevalence/existence of either Ixodes scapularis or Ixodes pacificus ticks, the bacterium (Borrelia burgdorferi spp), epidemiological data demonstrating ticks are infected, and population size/distribution in areas where people are found.
Very low risk areas, only I. scapularis ticks have been found, and there has been no record of infection with Borrelia burgdoferi identified.
- Oklahoma
- Kansas
- Oregon
- Florida
- Alabama
- Utah
- Georgia
- Texas
- Arkansas
- Nevada
- Arizona
- Missouri
- Mississippi
*Risk is calculated by prevalence/existence of either Ixodes scapularis or Ixodes pacificus ticks, the bacterium (Borrelia burgdorferi spp), epidemiological data demonstrating ticks are infected, and population size/distribution in areas where people are found.
No risk areas have either Ixodes ticks or Borrelia burgdorferi as resident species
- Alaska
- Colorado
- Hawaii
- Idaho
- Montana
- New Mexico
- Wyoming
*Risk is calculated by prevalence/existence of either Ixodes scapularis or Ixodes pacificus ticks, the bacterium (Borrelia burgdorferi spp), epidemiological data demonstrating ticks are infected, and population size/distribution in areas where people are found.
Who We Serve

General Population
At the American Lyme Disease Foundation, we are dedicated to providing up-to-date and credible information, dispel myths and misconceptions, and serve as a valuable resource for anyone with questions about Lyme disease and other tick-related illnesses. Trust us to provide the knowledge and support you need on your journey to better health.
Health Agencies
ALDF tackles the challenge of misinformation posted online, supporting health agencies to effectively manage risks and safeguard more patients. With our reliable information, we empower organizations to save lives by providing accurate and up-to-date resources. Together, we can make a difference.

Physicians and Healthcare Professionals
Doctors and healthcare professionals play a crucial role in combating misinformation about Lyme disease and tick-related illnesses. At ALDF, we serve as a trusted channel for patients to connect with the right healthcare professionals who can provide accurate information, diagnosis, and treatment. Research. Education. Prevention. That’s our commitment to you.
Looking for a specific topic?
Who We Are
Our Mission
Our goal is to provide up-to-date and credible information, dispel myths and misconceptions, and serve as a resource for anyone with questions about Lyme disease and other tick-related illnesses.
The Team
Our staff includes leading academic research scientists who have published more than 800 articles in top medical journals.
Awards and Certifications

Among websites specifically devoted to Lyme disease, ALDF ranks first for accurate Information related to Lyme Disease by the NIH National Center for Biotechnology Information

